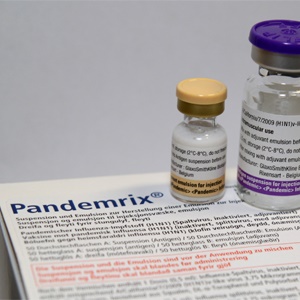
When a swine flu pandemic took the world by storm in 2009, drugmakers raced to prepare a vaccine capable of reducing the spread of the virus – but did this same drug cause an increase in narcolepsy amongst those who received it?
In April 2009, reports of a new flu virus began to emerge from Mexico. This new virus had a unique composition of flu genes never seen before in animals or humans, according to the US Centres for Disease Control and Prevention.
By the 15th of April 2009, the first U.S case was detected in California and shortly afterwards, further cases were reported in California, Texas and New York. The virus quickly gained momentum, with cases being reported more frequently in both Mexico and North America.




 Publications
Publications
 Partners
Partners