A landmark clinical trial has shown that a compound in marijuana can ease life-threatening seizures in children with a rare and devastating form of epilepsy.
Health24 previously reported that the Western Cape High Court has made a ruling allowing the possession, cultivation and usage of dagga in the home.
'Gold standard' evidence
Cannabidiol (CBD) – a non-intoxicating chemical – reduced seizure frequency by 39% in patients with Dravet Syndrome, researchers report.
The study appears in the New England Journal of Medicine.
This is the first randomised, controlled trial to show that CBD can help control seizures in some people with epilepsy, said study author Dr Orrin Devinsky. He is director of the Comprehensive Epilepsy Center at NYU Langone Medical Center in New York City.
"It's a big landmark in the scientific study of cannabis, and it's a major landmark in epilepsy care," Devinsky said. "After four millennia of using cannabis to treat epilepsy, we now have for the first time scientifically rigorously obtained data that this specific compound works in this specific form of epilepsy."
Brandy Fureman, vice president of research and new therapies for the Epilepsy Foundation, agreed that the new trial provides "gold standard" evidence of CBD's effectiveness.
Clinical trial
The clinical trial relied on a liquid formulation of CBD called Epidiolex, which was developed by British company GW Pharmaceuticals.
Epidiolex has not been approved by the US Food and Drug Administration. GW Pharmaceuticals – who paid for the clinical trial – expects to file for FDA approval of the drug this year.
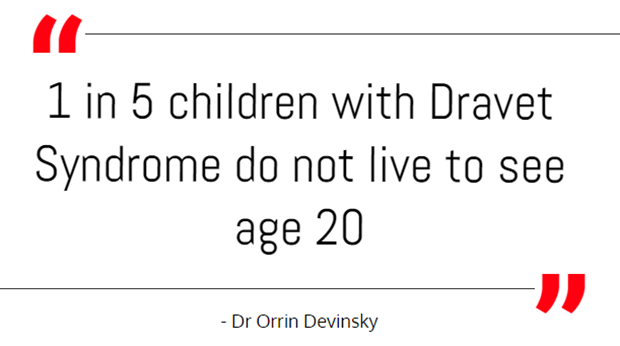
In the trial, Devinsky and his colleagues recruited 120 children and teenagers with Dravet Syndrome, which generally starts causing severe seizures within the first year of life. The seizures often are prolonged and repetitive; one in five children with Dravet Syndrome does not live to see age 20, Devinsky said.
Some seizures stopped entirely
The patients, ranging in age from two to 18, were randomly assigned to receive every day either 20 milligrams of liquid Epidiolex or a placebo, on top of their usual medication. The study took place across 23 sites in the United States and Europe, and lasted 14 weeks.
Children receiving Epidiolex experienced fewer seizures, going from an average of 12 convulsive seizures a month before the study to about six seizures a month. Three patients' seizures stopped entirely.
At the same time, children in the placebo group only had a slight reduction in seizures, from about 15 to 14 seizures a month.
"Like other epilepsy medications, CBD appears to work for some people and not for others," Fureman said.
Read more:
Epileptic girl becomes Mexico's first legal consumer of medical marijuana




 Publications
Publications
 Partners
Partners











