According to an article on centerwatch.com, currently less than 10% of the top listed pharmaceutical companies’ marketed products are biologic drugs, but these products nevertheless comprise 40% of these companies’ drug pipelines. Most of these pipeline biologicals focus on the treatment of cancer, infection and central nervous system diseases.
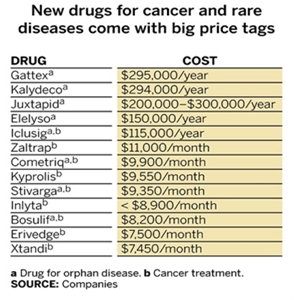
Peter Jordan, Principal Officer of Fedhealth, says it is interesting to note that new biologicals are not the only drugs which will lead to an increase in medicine costs. In 2012, 39 new drugs were approved by the FDA. These are some of them. The top 5 on this list are not biological drugs.
Of the nine new listed cancer treatments approved by the FDA last year, only two were biological – Erivedge, completely new in its class for the treatment of advanced skin cancer (basal-cell cancer), and Zaltrap, used in the treatment of advanced colorectal cancer.
For medical schemes it is always a major challenge to try and justify the cost of these new drugs compared against the patient benefit. Looking more closely at Zaltrap as an example - results from a main study showed that use of the drug came with a significant increase in problem side effects and more than double the number of patients in the Zaltrap trial actually dropped out of the study when compared to the patients only on standard therapy. By today’s exchange rate, the cost of Zaltrap is R101 880,00 per month ($11 000), for an average additional survival advantage of 1.44 months compared to standard chemotherapy.
“There are hundreds of other drugs in the pipeline which will have similarly high price tags,” he says.
So how are medical schemes dealing with the high cost of treatment? “While Fedhealth embraces true innovation, we agree with sentiments expressed by a group of cancer experts in a recent article in global publication, Blood. We believe drug prices should reflect objective measures of benefit, but should also not exceed values that harm our patients and societies,” says Jordan
Fedhealth’s managed care protocols are developed after extensive research is done involving careful analysis of trial data and consideration of international and local funding and clinical recommendations. “Critical questions asked when appraising a new drug include how much value it adds. This includes whether the new drug provides significant advances over existing therapies already funded or whether it offers advantages in terms of safety (fewer side-effects). If there are no appreciable advantages to the members being treated, or their families, the principles of cost-minimisation are applied i.e. the alternative therapy is funded.”
Along with this, actuarial analysis is done to determine the impact that funding of a new drug may have on contributions and how best to manage access by means of benefit design.
“Increasingly, the scheme is engaging with doctor networks in partnering to make decisions about the appropriate use of these expensive drugs. Where a new drug is funded by Fedhealth, managed health care programmes monitor whether the drug is used in the correct setting, as indicated, and conduct regular reviews to determine ongoing response. Where a new drug is not funded, or only funded by Fedhealth in restricted settings, engagement with the pharmaceutical companies has led, in some instances, to reductions in the South African single exit price,” Jordan explains.
An increase in healthcare spend on new drugs is inevitable, but, says Jordan, Fedhealth will continue to base funding decisions on scientific assessment of benefit versus harms, “while considering affordability, of course. We will also exert pressure, where possible, to strive toward value-based pricing of drugs,” he concludes.




 Publications
Publications
 Partners
Partners









